Table of Contents
Cell death
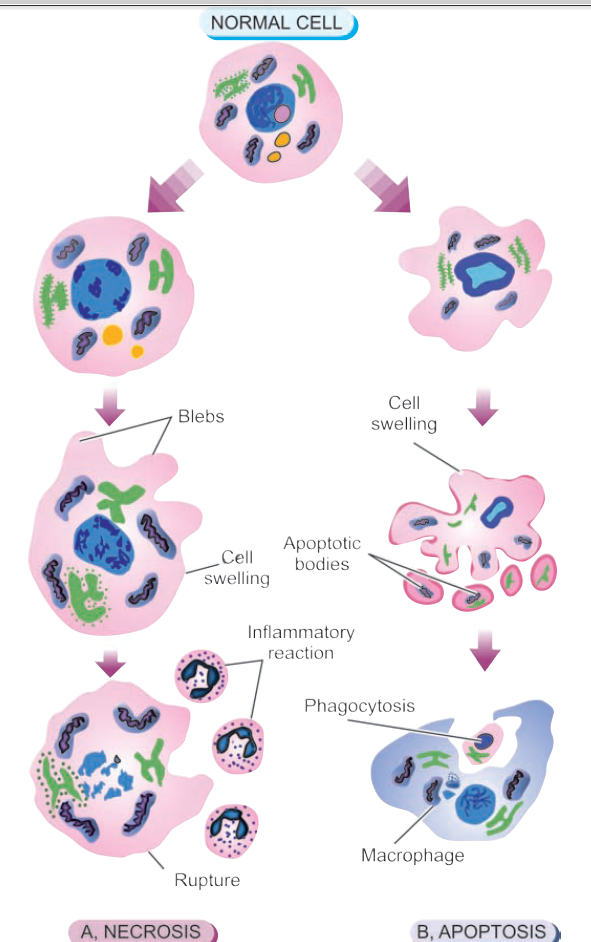
Cell death, the ultimate result of cell injury, is one of the most important events in pathology, affects every type of cell, and is the main result of ischemia (lack of blood flow), infections, toxins, and immune reactions. There are two main patterns of cell death: necrosis and apoptosis.
Necrosis
Necrosis is defined as a localized area of death of tissue followed by degradation of tissue by hydrolytic enzymes liberated from dead cells; it is invariably accompanied by an inflammatory reaction. Necrosis can be caused by various agents such as hypoxia, chemical and physical agents, microbial agents, immunological injury, etc. Two essential changes characterize irreversible cell injury in necrosis of all types :
i) Cell digestion by lytic enzymes. Morphologically this change is identified as homogeneous and intensely eosinophilic cytoplasm. Occasionally, it may show
cytoplasmic vacuolation or dystrophic calcification.
ii) Denaturation of proteins. This process is morphologically seen as characteristic nuclear changes in the necrotic cells. These nuclear changes may include condensation of nuclear chromatin (pyknosis) which may either undergo dissolution (karyolysis) or fragmentation into many granular clumps (karyorrhexis).
Types of Necrosis
Morphologically, there are five types of necrosis: coagulative, liquefaction (colliquative), caseous, fat, and fibrinoid necrosis.
1. COAGULATIVE NECROSIS.
This is the most common type of necrosis caused by irreversible focal injury, mostly from sudden cessation of blood flow (ischemia), and less often from bacterial and chemical agents. The organs commonly affected are the heart, kidney, and spleen.
Grossly, foci of coagulative necrosis in the early stage are pale, firm, and slightly swollen. With progression, they become more yellowish, softer, and shrunken.
Microscopically, the hallmark of coagulative necrosis is the conversion of normal cells into their ‘tombstones’ i.e. outlines of the cells are retained so that the cell type can still be recognized but their cytoplasmic and nuclear details are lost. The necrosed cells are swollen and appear more eosinophilic than normal, along with nuclear changes described above. But cell digestion and liquefaction fail to occur (c.f. liquefaction necrosis). Eventually, the necrosed focus is infiltrated by inflammatory cells, and the dead cells are phagocytosed leaving granular debris and fragments of cells.
2. LIQUEFACTION (COLLIQUATIVE) NECROSIS
Liquefaction or colliquative necrosis occurs commonly due to ischaemic injury and bacterial or fungal infections. It occurs due to the degradation of tissue by the action of powerful hydrolytic enzymes. The common examples are infarct brain and abscess cavity.
Grossly, the affected area is soft with a liquefied center containing necrotic debris. Later, a cyst wall is formed.
Microscopically, the cystic space contains necrotic cell debris and macrophages filled with phagocytosed material. The cyst wall is formed by proliferating capillaries, inflammatory cells, and gliosis (proliferating glial cells) in the case of the brain and proliferating fibroblasts in the case of the abscess cavity.
3. CASEOUS NECROSIS
Caseous necrosis is found in the center of foci of tuberculous infections. It combines features of both coagulative and liquefactive necrosis.
Grossly, foci of caseous necrosis, as the name implies, resemble dry cheese and are soft, granular, and yellowish. This appearance is partly attributed to the cytotoxic effects of lipopolysaccharides present in the capsule of the tubercle bacilli, Mycobacterium tuberculosis.
Microscopically, the necrosed foci are structureless, eosinophilic, and contain granular debris. The surrounding tissue shows a characteristic granulomatous inflammatory reaction consisting of epithelioid cells with interspersed giant cells of Langhans’ or foreign body type and peripheral mantle of lymphocytes.
4. FAT NECROSIS
Fat necrosis is a special form of cell death occurring at two anatomically different locations but morphologically similar lesions. These are: following acute pancreatic necrosis, and traumatic fat necrosis commonly in breasts. In the case of the pancreas, there is the liberation of pancreatic lipases from injured or inflamed tissue that results in necrosis of the pancreas as well as of the fat depots throughout the peritoneal cavity, and sometimes, even affecting the extra-abdominal adipose tissue. Fat necrosis hydrolyses neutral fat present in adipose cells into glycerol and free fatty acids. The damaged adipose cells assume a cloudy appearance. The leaked out free fatty acids complex with calcium to form calcium soaps (saponification) discussed later under dystrophic calcification.
Grossly, fat necrosis appears as yellowish-white and firm deposits. The formation of calcium soaps imparts the necrosed foci firmer and chalky white appearance.
Microscopically, the necrosed fat cells have a cloudy appearance and are surrounded by an inflammatory reaction. The formation of calcium soaps is identified in the tissue sections as amorphous, granular, and basophilic material.
5. FIBRINOID NECROSIS
Fibrinoid necrosis is characterized by the deposition of fibrin-like material which has the staining properties of fibrin. It is encountered in various examples of immunologic tissue injury (e.g. in immune complex vasculitis, autoimmune diseases, Arthus reaction, etc), arterioles in hypertension, peptic ulcer, etc.
Microscopically, fibrinoid necrosis is identified by brightly eosinophilic, hyaline-like deposition in the vessel wall. Necrotic focus is surrounded by nuclear debris of neutrophils (leucocytoclasis). Local hemorrhage may occur due to rupture of the blood vessel.
Apoptosis
Apoptosis occurs when a cell dies through activation of an internally controlled ‘suicide’ program. After this, the dead cells are removed. Apoptosis is designed to eliminate unwanted cells during the development of the embryo and in various physiological processes. It also occurs under pathological conditions, where it is sometimes accompanied by necrosis.
The term was first introduced in 1972 as distinct from necrosis by being a form of cell death that is controlled and regulated by the rate of cell division; when the cell is not needed, the pathway of cell death is activated (‘cell suicide’) and is unaccompanied by any inflammation and collateral tissue damage.
APOPTOSIS IN BIOLOGIC PROCESSES
Apoptosis is responsible for mediating cell death in a wide variety of physiologic and pathologic processes as under:
Physiologic Processes
a. Organized cell destruction in the sculpting of tissues during the development of the embryo.
b. Physiologic involution of cells in hormone-dependent tissues e.g. endometrial shedding, regression of lactating breast after withdrawal of breastfeeding.
c. Normal cell destruction followed by replacement proliferation such as in intestinal epithelium.
d. Involution of the thymus at an early age.
Pathologic Processes:
a. Cell death in tumors exposed to chemotherapeutic agents.
b. Cell death by cytotoxic T cells in immune mechanisms such as in graft-versus-host disease and rejection reactions.
c. Progressive depletion of CD4+T cells in the pathogenesis of AIDS.
d. Cell death in viral infections e.g. formation of Councilman bodies in viral hepatitis.
e. Pathologic atrophy of organs and tissues on withdrawal of stimuli e.g. prostatic atrophy after orchiectomy, atrophy of kidney or salivary gland on obstruction of ureter or ducts, respectively.
f. Cell death in response to injurious agents involved in the causation of necrosis e.g. radiation, hypoxia, and mild thermal injury.
g. In degenerative diseases of CNS e.g. in Alzheimer’s disease, Parkinson’s disease, and chronic infective dementias.
h. Heart diseases e.g. heart failure, acute myocardial infarction (20% necrosis and 80% apoptosis).
MORPHOLOGIC FEATURES
The characteristic morphologic changes in apoptosis seen in histologic and electron microscopic examination are
a. Involvement of single cells or small clusters of cells in the background of the viable cells.
b. The apoptotic cells are round to oval shrunken masses of intensely eosinophilic cytoplasm (mummified cell) containing shrunken or almost-normal organelles.
c. The nuclear chromatin is condensed or fragmented (pyknosis or karyorrhexis).
d. The cell membrane may show convolutions or projections on the surface.
e. There may be the formation of membrane-bound near-spherical bodies on or around the cell called apoptotic bodies containing compacted organelles.
f. Characteristically, unlike necrosis, there is no acute inflammatory reaction around apoptosis.
g. Phagocytosis of apoptotic bodies by macrophages takes place at varying speeds. There may be swift phagocytosis, or loosely floating apoptotic cells after losing contact, with each other and basement membrane as single cells, or may result in major cell loss in the tissue without significant change in the overall tissue structure.